Category: Uncategorized
-

Dog Down Syndrome: Symptoms, Causes, and Diagnosis
Continue ReadingDog Down Syndrome
Many disorders are common in all organisms sustaining in this world. Any change in structure or number of an organism result in genetic disorders, as a result of these mutations many severe symptoms occur which leads to a death of an individual.
These syndromes are also common other animals as of humans. Downs syndrome is a condition where there is addition of one number of chromosomes in the cell. It often occurs in chromosome number 21.
Does Dogs Have Down Syndromes?
Yes! definitely; each and every living organism even plant is supposed to many diseases and all of us take medications and treatment as all, we want is to have a healthier life.
Before knowing whether dogs have this syndrome it is necessary to understand about down syndrome.
The addition or extra number of chromosomes present in the chromosome number 21 causes down syndrome.
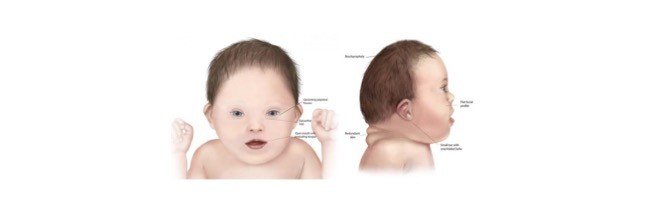
In humans, it is generally categorized by flattened nose, protruded tongue and mental retardation. Where as they also suffer with other disorders like hearing loss, or eye diseases like cataract.
As given above there are so many ways to find a Down syndrome in humans but it is not so easy to find whether the dog is suffering from any syndrome unless any other syndromes show the physical characteristics.
We all know that the number of chromosomes present in the cells differ accordingly with each species, where we; the humans contain 23 pairs of chromosomes but dogs contain 39 pairs of chromosomes.
Downs syndrome in humans can be diagnosed by having 47 number of chromosomes but where as the dogs contains 78 number of chromosomes.
Dog Down Syndrome Diagnosis
These kind of chromosomal abnormalities leads to early death of dogs. Usually, genetic tests are followed to identify this syndrome. But usually, this condition does not truly exist in dogs.
Does Dog Down syndrome Really Exist?
Downs syndrome does not truly exist in dogs. On the other hand, other congenital and developmental conditions occur in dogs where the symptoms are similar to that of downs syndrome, Congenital hypothyroidism is one of the good examples of this type of congenital disease.
Some of the symptoms that resemble as such of down syndrome is slow growth which eventually results in small stature, protrusion of tongue and also in large tongue than usual.
Short limbs, poor muscle tone, delaying in opening of eyes and ears etc.
The other symptoms which relate the congenital diseases with downs syndrome is pituitary dwarfism, congenital hypo Cephalus, growth hormone deficiencies and portosystemic shunt.
Symptoms That Mimic Dog Down Syndrome
If we find a dog by struggling with growth delays or any other cognitive diseases, apart from genetic diseases there is chances of having other congenital heart or thyroid issue which are due to pituitary dwarfism or any other growth hormone deficiencies where all these disorders and symptoms that results as such of down syndrome.
Dog Down Syndrome Symptoms
Along with above said symptoms the other symptoms that doubts whether it is a down symptom are as follows.
Poor Eye Sight: Hence in this condition dogs have poor health conditions which results in poor eyesight. They also have ahigh risk of developing cataract at least in any one of the eyes. In this case the cloudiness of the eyes should be checked with Veterinarian. Because this condition is the best symptom of forming a cataract.
Unusual Physical Features: Normally dogs with this condition have different facial features and shortened growth. Other common symptoms include shortened neck, flattened face, small structure of head, slanting eyes with a flap of skin above the eyes. And ears are abnormal in structure. Along with these, the affected dogs also have dry noses, patches on skin, incomplete legs, and they start shedding their hair from the fur.
Hearing Deficiencies: The affected dogs also have the hearing problems but which is very difficult to diagnose, because the dog having these conditions follow the commands later and they are always slow in reacting to the situations.
Pain: The dog with these symptoms usually gets it whole body affected which also results in abnormal circulatory system and causes severe and random pains throughout the body of the dog,
Unusual Discharges: These dogs usually with this condition has many diseases along with this congenital disease which results in abnormal blood discharge from their rectum. We can also notice this discharge coming out randomly and often during the later stages of their infection.
Skin Problems: We can see many dogs along the streets that are suffering from skin disease. But the dogs with these associated symptoms forms missing and abnormal patches and causes irritant to the skin and it also led to other health problems.
Thyroid Problems: Dogs with these down syndromic symptoms are usually associated with thyroid problems due to malfunctioning of the thyroid gland, which results in metabolic problems and other effects in controlling the temperature of our body. Generally, the dogs with these symptoms have low temperature when compared to normal healthy dogs.
Behavioral Issues: Dogs who are suffering with this syndrome show odd behaviors, howling or wailing unnecessarily than usual times. So, it finds very difficult to feed and care like other usual dogs. They also cannot control their bladders as well. So, they feel very discomfort and howl deeply.
However, it is still not yet confirmed that downs syndrome is also present in dogs.
Care to be Taken
The dog showing all these symptoms can be taken for a veterinarian regularly once in a week and they had to given with proper medications.
They should also be taken for a walk twice in a day. It is also important to observe the emotional and social skills of the dog.
It is also vital to keep in touch with their body languages and to find out the emotion that they want to conquer us.
However, the other symptoms associated with down syndrome with the dogs leads them to death, in many cases it is said that mother itself will kill the dog which is having abnormality.
Or in other cases the mother dog also isolates the affected puppy to prevent the infection passing through other puppies through the affected one’s litter.
But mostly this condition is spelled out as down syndrome because it is till now not clear whether dogs obtain down syndrome.
Down Syndrome Citations
Share
Similar Post:
-

Metformin: Description, Properties, and Health Benefits
Continue ReadingHistory of Metformin
Recently called metformin, Dimethylbiguanide, was initially integrated in 1929 after an investigation into a more harmful hypoglycaemic subsidiary guanidine; found in Galega officinalis which had been utilized to treat the sign and effects of diabetes since the seventeenth century.
Metformin was not utilized in Europe to treat diabetes, because of the poisonousness of its subsidiaries and the creation of engineered insulin in 1922, until the last part of the 1950’s when French Physician Jean Sterne who made note of its antihyperglycaemic impacts during preliminaries into discovering new malarial medicines.
Metformin and Type-II Diabetes
Today, metformin is the principal line and most ordinarily endorsed prescription for the treatment of type-II diabetes mellitus (T2DM).
This is as it is unfathomably protected, savvy, being one of the fewanti-hyperglycaemic prescriptions to not build weight and decrease cardiovascular sickness hazard.
In spite of metformin’s fame, there is as yet a general absence of comprehension of its systems of activity.
Metformin likewise has low power, with a maximum portion of 2500mg, and regular contraindications coming about in around 25% of medicines.
Does this suggest the conversation starter; is metformin the best first-line treatment for type-II diabetes mellitus?
Mechanism of Metformin
Since the 1950’s there have been considerable endeavors into understanding the manners by which metformin can bring down hepatic glucose creation (HGP) and plasma glucose levels (PGL).
Up to this point, the hypothesis was that metformin’s enemy of hyperglycemic impact was fundamentally intervened through a Liver Kinase B1 (LKB1) – 5′ AMP-enacted protein kinase (AMPK) subordinate pathway.

This has now been tested after examinations showing that hereditarily inadequate mice, lacking AMPK in the liver, kept up with tantamount blood glucose levels to that of the benchmark group of mice.
This shows that metformin doesn’t intervene gluconeogenesis, essentially exclusively, in the liver through the LKB1-AMPK pathway.
Subsequently further exploration has been embraced to decide the genuine instruments of activity of metformin.
There are presently different working speculations concerning the objective for metformin’s capacity to oversee HGP notwithstanding almost certainly, it intervenes through various methods of activity.
One late hypothesis is that, because of high measures of metformin should have been powerful, it adjusts the microbiota of the host, expanding Akkermansia muciniphila populaces.
A. muciniphila was displayed to altogether expand glucose resistance in High-fat-diet (HFD) prompted diabetic mice.
There is likewise mounting proof from contemplates showing the gut may assume a vital part in metformin’s intervention of HGP and PGL. One such examination showed that intraduodenal mixtures of metformin gave the main change in HGP.
This examination likewise exhibited the dependence of glucagon-like peptide-1 receptors (GLP-1R) and protein kinase A (Pka) to bring down HGP, recommending an AMPK – 1.
GLP-1R – Pka subordinate pathway. This signallling pathway doubtlessly brings down HGP through neuronal intercession of glucose homeostasis through the gut-cerebrum liver hub.
Different investigations have likewise shown supplement detecting upper intestinal lipids setting off a gut-cerebrum liver pivot to direct gluconeogenesis exhibiting the body’s capacity to control through this pathway.
Advantage of Metformin in Diabetes
Metformin is used for treatment of Type 2 Diabetes Mellitus, this is to a great extent because of its expense, solid wellbeing record, minor incidental effects – discarding uncommon instances of lactic acidosis – and cardioprotective impacts.
Metformin might be the best first-line treatment for overweight type II diabetic patients.
Another advantage of metformin over other regular enemy of hyperglycaemic medicines is that it doesn’t bring about extra weight acquire and may advance less eating and weight reduction.
This is probably because of its capacity to decrease HGP giving less glucose in the circulation system for adipocytes to change over into fat contrasted with insulin which would have the contrary impact and increment weight acquire because of expanded cell take-up.
Is Metformin really Effective?
Metformin tends to the manifestations of hyperglycaemia as opposed to effectively remedying the basic issues related with T2DM.
Diabetes is regularly described to have 8 significant physiological disturbances known as the ominous octet.
Along these lines thought to have a powerful treatment in which HbA1c is brought down and kept up with by turning around the fundamental dysfunctions related with the infection.
One such proposed treatment is regulating the GLP-1R agonists (GLP-1RAs). GLP-1RAs, for example, liraglutide have exhibited the capacity to address 6 of the ominous octets.
It has likewise been shown that liraglutide brings down HbA1c quicker and drastically more than metformin, a decrease of 1.14% (1.8mg-liraglutide) longer than a year, contrasted with sulfonylurea, which has comparative HbA1c decrease to metformin, lessening by 0.51%.
Hence thought to have a powerful treatment in which HbA1c is brought down and kept up with by switching the fundamental dysfunctions related with the infection.
One such proposed treatment is regulate GLP-1R agonists (GLP-1RAs). GLP-1RAs, for example, liraglutide have exhibited the capacity to address 6 of the ominous octet.
It has additionally been shown that liraglutide brings down HbA1c quicker and drastically more than metformin, a decrease of 1.14% (1.8mg-liraglutide) longer than a year, contrasted with sulfonylurea, which has a comparative HbA1c decrease to metformin, lessening by 0.51%.
Metformin has obviously solid advantages related with treatment. It is reliably viable in decreasing danger factors related with T2DM and is adequate treatment in lessening both HGP and PLG.
Nonetheless, because of the far and wide utilization of metformin and its evident adequacy different medicines, for example, GLP-1RAs are not being investigated as generally accessible choices for patients with T2DM.
It ought to likewise be noticed that regardless of the clear suitability of GLP-1RAs, there is yet to be a moderate choice, liraglutide yearly expense is ~$9,300 contrasted with metformin <$50.
Further exploration should likewise be done into the conceivable results of GLP-1RAs as there is still some debate encompassing expanded dangers of the thyroid malignant growth, pancreatitis and the kidney impairment.
Considering the expenses and the vulnerability encompassing more current choices, for example, GLP-1RAs I accept that metformin ought to stay the main line treatment for type-II diabetes mellitus in any case, further investigation into new strategies following the very authoritative opinion as that of GLP-1RA medicines ought to be investigated in order to track down a more successful and designated treatment.
Metformin Citations
Share
Similar Post:
-
Cell Cycle Assay With Flow Cytometry: Detailed...
Continue ReadingAbout PhD In Psychology
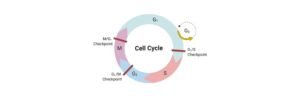
Cell cycle analysis by quantitation of DNA content was one of the fantastic applications of flow cytometry.
The mammalian DNA, yeast, DNA, plant DNA or bacterial DNA can be easily stained by a variety of DNA binding dyes such as propidium iodide.
The premise of these DNA binding dyes is that they are stoichiometric, i.e. they bind in proportion to the DNA amount present in the cell.
In this way if cells are in S phase will have more DNA than cells in G1 phase.
They will take up proportionally more dye and will fluoresce more brightly until they have doubled their DNA content.
The cells in G2 phase will be approximately twice as bright as cells in G1. DNA-binding dyes include propidium iodide (PI), Hoechst 33342, 33258 and S769121, 7-aminoactinomycin-D (7-AAD), DRAQ5™ and DRAQ7™, TO-PRO-3, 4’6’-diamidino-2-phenylindole (DAPI).
In general, cells must be fixed and permeabilized to allow entry of the DNA binding dye which is otherwise actively pumped out by living cells.
Alcohol or aldehyde are commonly used to fix the cells.
Alcohol is a dehydrating fixative which also permeabilizes cells of interest. This will allow easy access of the dye to the DNA and gives good profiles (low coefficient of variation, CV).
The disadvantage of alcohol fixation is that it is often incompatible with fluorescent proteins and some surface markers.
For simultaneously proteins or surface markers examination, use of an aldehyde (cross-linking) fixative, such as paraformaldehyde is more efficient and appropriate.
Aldehyde fixation in cell cycle analysis may lead to poorer quality profiles (higher CVs) but will allow simultaneous detection of other fluorochromes and membrane-bound surface proteins.
Another disadvantage of paraformaldehyde fixation is, generally paraformaldehyde does not permeabilize the cell membrane, and so further sample processing is required.
With fixed cells, samples may be processed, stained and analyzed at the conclusion of an experiment.
Alcohol-fixed cells are stable for several weeks at 4°C and several weeks in -20 °C.
Aldehyde fixed cells are stable for 2 to 3 days. An alternative method to allow the DNA dye into the cells is to permeabilize them with a detergent such as Triton X-100 (0.1%) or NP40 (0.1%).
Usually, it is also necessary to combine a fixation (paraformaldehyde) and permeabilization (Triton X-100) for the intracellular staining.
On the other hand, methods are available, e.g. use of citrate buffers (in combination with detergent), although these are not so widely used.
There are also some dyes that will enter live cells and quantitatively bind to DNA, these include Hoechst 33342, DRAQ5™ (ab108410) and the DyeCycle dyes
Adopted from BioRender
Cell Cycle Assay Reagents
70% Ethanol
Propidium iodide (stock solution 50 µg/ml)
Ribonuclease I (stock 100 µg/ml)
Cell Cycle Assay Procedure
1. Seed appropriate number of cells (e.g. 1.5×105 MDA-MB-231, 1X105 MCF-7, and 1X105 SK-BR-3) in 6 well plates in 2 ml complete media.
Seed less number of cells in control wells if your cells are fast growing like MDA-MB-231 so that they will not die due to over confluence.
Incubate in 5% CO2 incubator for 18-24 hours.
2. When the cells are at 40-50% confluence, discard the media and add 1 ml of serum deprived media (containing 0.5% FBS) to synchronize the cells.
Incubate in 5% CO2 incubator for 6 hours.
3. After this period discard the media and add 1 ml complete media.
Then, treat the cells in same media at different doses of your test compound in triplicates. Make a negative control with no treatment.
Incubate in 5% CO2 incubator for 18-24 hours.
4. Harvest the cells (both dead and live cells) after the incubation period by trypsinizing them (add 500 μL Trypsin per well).
Pool triplicate wells into single 15 ml tube.
Centrifuge at 1000 rpm for 5 minutes to pellet down.
5. Wash the pelleted cells with 2-3 ml of PBS.
Re-centrifuge the cells at 1000 rpm for 5 minutes.
6. Give second washing with 2-3 ml PBS if necessary.
Again centrifuge cells at 1000 rpm for 5 minutes.
7. Discard the PBS. Gently mix the pelleted cells in the remaining PBS and fix each cell suspension in 1-3 ml of 70% ethanol (depending on the number of cells in the suspension).
8. Keep them at -200C for a minimum of 1 day (although they can stay for up to 2-3 days).
9. Spin down the cells at 1800 rpm for 5 minutes.
Discard the supernatant.
Wash the pellet in 2-3 ml PBS.
Again spin down at 1800 rpm for 5 minutes.
10. Give a second wash with PBS if necessary.
Spin down the cells at 1800 rpm for 5 minutes to remove the traces of ethanol.
Mix the cell pellet in remaining PBS.
11. Make PI-RNase master mix by taking 5μl of PI (2mg/ml stock) and 2μl of RNase (10mg/ml stock) in 300 μl PBS per sample.
12. Add 300 μl of master mix per sample. Mix well.
13. Incubate for 30 minutes in dark at RT.
Transfer to FACS flow tubes. Keep the tubes on ice.
14. Analyse the samples via FACS as soon as possible and interpret using software
Cell Cycle Assay Citations:
Share
Similar Post:
-

Mycobacterium Tuberculosis : A Complete Overview I...
Continue ReadingWhat is Tuberculosis
Tuberculosis (TB), which is caused by Mycobacterium Tuberculosis, one of the oldest recorded human afflictions, is still one of the biggest killers among the infectious diseases.
Apart from this, TB is the most common cause of death in human immunodeficiency virus (HIV) positive patients, since both (TB &HIV) are more destructive together than either alone.
A total of 30 million people worldwide are infected with HIV and one third of them are co-infected with TB, which account for up to ~33% mortality per year.
Furthermore, the prevalence of multi-drug-resistance (MDR) TB in HIV infected patients, have rises up to 9%, since the last global drug resistance survey.
The global threat of XDR tuberculosis has great significance for the public health field. For one thing, its very existence is a reflection of weaknesses in tuberculosis management, which should minimize the emergence of drug resistance.
"A total of 30 million people worldwide are infected with HIV and one third of them are co-infected with Mycobacterium tuberculosis, which account for up to ~33% mortality per year"
What is Mycobacterium tuberculosis
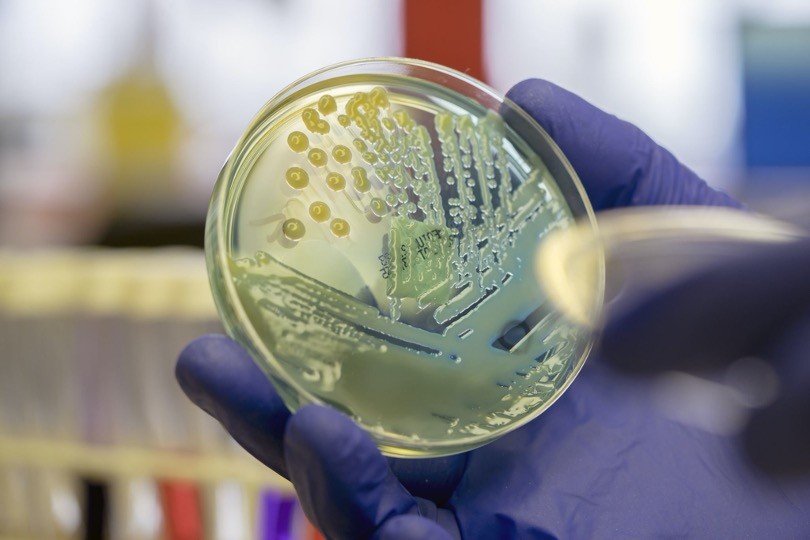
Mycobacterium tuberculosis are rod or slightly curved rod, approximately 1-10 µm long and 0.3-0.6 µm in diameter, occurring singly, in pair or as small clumps.
Mycobacterium tuberculosis is a slow-growing, facultative intracellular pathogen that can survive and multiply inside macrophages and other mammalian cells.
It is non-motile, non-spore forming, aerobic and weakly gram positive. It shares number of genes with other members of the Mycobacterium genus.
It has a unique cell wall, due to the dominant presence of mycolic acids that make up more than 50% of its dry weight. It is distinguished from other bacilli by its acid fastness upon Zeihl Neelsen staining.
How Mycobacterium tuberculosis grows in culture?
The bacilli grow slowly with a generation time of 15-20 hours in vitro. The colonies appear in about three weeks and may some times takes up to eight weeks.

The optimum temperature for the growth of M. tuberculosis is 37°C with the optimum pH 6.4-7.0 and growth does not occur below 25°C or above 40°C.
Several solid and liquid media have been defined for the cultivation of M. tuberculosis bacilli.
The solid media most widely used for routine culture is Lowenstein Jensen (L-J) media and Middle brook 7H10 and Middle brook 7H9 for drug susceptibility testing (Recommended by the International Union against Tuberculosis).
On solid media, M. tuberculosis form opaque, dry colonies with a wrinkled or nodular surface. Colonies are creamy white, becoming yellowish or buff colored on further incubation, while in liquid media M. tuberculosis grows in the bottom and form serpentine cords or duster.
Global epidemiology of Mycobacterium tuberculosis
According to WHO surveys (2006) of the effectiveness of surveillance system, and death registration, there were an estimated 8.9 million new cases of TB in 2004. Based on data from 175 countries, the prevalence of MDR among new cases and among the previously treated cases are 3.1% and 19.3%, respectively.
When including other ten countries which reported data on combined drug resistance only, the global prevalence of MDR in all cases of TB has been reported to be 4.6%.
"XDR-TB is more expensive and difficult to treat than MDR-TB and outcomes for patients are worse, therefore understanding the magnitude and distribution of XDR-TB is important"

XDR-TB is more expensive and difficult to treat than MDR-TB and outcomes for patients are worse, therefore understanding the magnitude and distribution of XDR-TB is important.
Data from this report indicate that XDR-TB is widespread with 45 countries having reported at least one case.
The high proportions of XDR-TB among MDR-TB as well as the large overall burden suggest a significant problem within the countries of the Soviet Union.
Japan and republic of Korea in a previous study, has also shown a high proportion of XDR-TB among MDR.
South Africa reported a moderate proportion of XDR-TB among MDR-TB cases.
However, the underlying burden of MDR-TB is considerable and 44% of TB patients are estimated to be co-infected with HIV (WHO Report, 2008).
Among 17,690 M. tuberculosis isolates included in the WHO surveillance for drug resistant M. Tuberculosis in the network of TB laboratories 20% were MDR and 2% XDR.
In addition, population based data on drug susceptibilities of M .tuberculosis isolates obtained from USA (for 1993-2004), Latvia (for 2000-2002) and South Korea (2004) shows that 4%, 19% and 15% of MDR-TB cases, respectively, could be classified as XDR-TB.
Thus, the distribution of XDR-TB has been found to be worldwide (WHO Report, 2008).
Classification of Mycobacterium tuberculosis
Mycobacterium tuberculosis belongs to family Mycobacteriaceae and it is the only genus in this family.
The majority of the species that comprise the genus Mycobacterium are non-pathogenic environmental bacteria related closely to the soil bacteria Streptomyces and Actinomyces.
However a few species are highly pathogenic, including M. tuberculosis complex, M. leprae, and M. ulcerans, the causative agents of tuberculosis, leprosy and Buruli ulcers, respectively.
"The Mycobacterium tuberculosis complex consists of M. tuberculosis, along with M. bovis, M. canettii, M. africanum, and M. microti"
The Mycobacterium tuberculosis complex consists of M. tuberculosis, along with M. bovis, M. canettii, M. africanum, and M. microti, which are closely related organisms (share >99% identity at the nucleotide level for some loci) and are pathogenic.
However, they differ significantly in morphology, biochemistry, host range, and disease patterns in experimental animals.
Mycobacterium tuberculosis is the causative agent of tuberculosis in humans, although in part of Africa, M. africanum causes more cases of tuberculosis than M. tuberculosis.
In contrast, infection by M. canettii appears to be rare. M. bovis has a broad host range, including humans and cattle, and was a major cause of human tuberculosis prior to pasteurization of milk. M. microti is a pathogen of voles but is avirulent in humans and mice.
Evolution of Mycobacterium tuberculosis
The recent studies into the evolution of Mycobacterium tuberculosis have indicated that Mycobacterium tuberculosis is actually more closely related to a common founder strain that is M. bovis. This implies that either humans infected cattle with M. tuberculosis, resulting in the divergence of M. bovis, or the two strains evolved in parallel from a founder strain that infected both humans and cattle.
The ancestor of M. tuberculosis most likely arose from the M. tuberculosis-like species of the M. tuberculosis complex that are found today in central Africa.
This might indicate that humans were exposed to tuberculosis-mediated selection pressure much earlier, and more comprehensively, than was previously assumed. However, M. africanum and M. microti represent intermediates in this new evolutionary scenario.
Natural Habitats of Mycobacterium tuberculosis
The genus mycobacterium includes obligate parasites, saprophytes and opportunistic pathogens. Most of the species are free living in soil and water, but the major ecological niche for other such as M. tuberculosis complex and M. leprae is diseased tissues of human and other warm-blooded animals.
Cell wall composition of Mycobacterium tuberculosis
M. tuberculosis possesses a polysaccharide cell wall that resembles gram positive bacteria in having disaccharide linker between peptidoglycan and polysaccharide.
The cell wall of M. tuberculosis contains 3 classes of mycolic acids: α-, keto-, and methoxymycolates. α-Mycolic acid has 2 cyclopropane rings, which are in the cis configuration, while the keto- and methoxymycolates have1 ring each in either cis or trans configuration.
In addition to trehalose, mycolates are linked to arabinogalactan which is covalently attached to the peptidoglycan layer. The pathogenic strains of M. tuberculosis posses “cord factor”, which are highly toxic in mice.
"The cell wall of M. tuberculosis contains 3 classes of mycolic acids: α-, keto-, and methoxymycolates. α-Mycolic acid has 2 cyclopropane rings"
Recent studies suggest that cord factor is made up of trehalose dimycolate (TDM), which are composed of a non-reducing sugar trehalose linked to mycolic acids.
Share
-

Types of Breast Cancer : Non-Invasive, Invasive...
Continue Reading"Breast cancer now represents the most common female malignancy in both the developing and developed world, and is the primary cause of death among women globally"
Epidemiology Of Breast Cancer
Breast cancer is most commonly diagnosed malignancy among women worldwide.
Breast cancer now represents the most common female malignancy in both the developing and developed world, and is the primary cause of death among women globally.
Breast cancer is the second most common cancer with 1.7 million cases (11.9%) reported in 2012, more cases occurring in less developed (883,000 cases) than more developed regions (794,000) worldwide.
Since the 2008 estimates, breast cancer incidence has increased by more than 20%.
It now represents one in four of all cancers in women, while mortality has increased by 14%.
"Breast cancer ranks as the fifth cause of death from overall cancers"
Breast cancer ranks as the fifth cause of death from overall cancers (522,000 deaths, 6.4%) and while it is the most frequent cause of cancer death in women in less developed regions (324,000 deaths, 14.3% of total), it is now the second cause of cancer death in more developed regions (198,000 deaths, 15.4%) after lung cancer.
The incidence of breast cancer dramatically increases from 1971-2010 due to change in life style habits. 1 woman out of 8 is at the risk of getting breast cancer.
In India breast cancer ranked second cancer after cervical cancer. Estimated number of breast cancer cases in women in 2012 was 144,937 (27%) in India.
Different Breast Cancer Types
Breast cancer is a heterogonous disease with different molecular profiles. Different morphological features arises as result of different genetic, epigenetic and transcriptomic alterations in mammary epithelial and stromal cells.
Breast cancer is the carcinoma mammary epithelial cells which lined lobules or milk duct. Depending upon the site of origin cancer cells breast cancer of two type lobular carcinoma, carcinoma of the lobular epithelial cells and ductal carcinoma originate from the epithelial cells which lined the milk duct.

Both ductal and lobular carcinomas are further sub classified as invasive and non invasive carcinoma.
Non-invasive carcinoma in which cancer cells still confine to their parental origin site is called ductal or lobular carcinoma in situ.
When cancer cells are spread to surrounding tissue from their parental origin site is called invasive carcinoma.
"Breast cancer is a heterogenous disease with different molecular profiles"
Non invasive carcinoma of two types depending on their origin site as follows:
Ductal carcinoma in situ is a non invasive carcinoma of milk duct epithelial cells which still confined to their parental origin and does not have the potential to metastases.
If not treated DCIS result in the invasive tumor which are difficult to manage.
DCIS range from low grade to high grade tumor including intermediate grade.
Early detection symptoms are lump in the breast fluid discharge from the nipple and rashes on the nipple on breast.
"Ductal carcinoma in situ (DCIS), which accounts for 65,00 (20–30 %) of breast cancers each year"
Ductal carcinoma in situ (DCIS), which accounts for 65,00 (20–30 %) of breast cancers each year and is most commonly diagnosed on screening mammography, traditionally has been managed through a combination of surgery, radiation therapy (RT), and endocrine therapy.
Loss of heterozygosity is noted in more than 70 percent of high-grade ductal carcinomas in situ, as compared with 35 to 40 percent of cases of atypical hyperplasia and 0 percent in specimens of normal breast tissue.
Lobular carcinoma in situ (LCIS) is a carcinoma of the lobule, milk producing gland at the ends of breast duct, which still confined to their parent origin site in lobules is called lobular carcinoma in situ.
Lobular carcinoma in situ (LCIS) is considered to be a risk factor for the development of invasive breast carcinoma, but it may also be a non-obligate precursor to invasive lobular carcinoma (ILC).
LCIS is believed to arise from atypical lobular hyperplasia (ALH).
LCIS is most common among the menopausal women and these women are at 8 to 10 times more risk developing the invasive breast cancer.

Invasive ductal carcinoma is the most common type of breast cancer sometimes called infiltrating ductal carcinoma of the milk duct.
This is the largest group of malignant mammary tumors, comprising 75% to 80% of mammary carcinomas.
With invasive cancer, cancer cells start in a milk duct, break through the duct walls, and then invade fatty breast tissue.
Invasive cancer can remain localized, or the cancer cells may enter the bloodstream or lymphatic system and metastasize.
Invasive ductal carcinoma is the most common type of invasive breast cancer and accounts for 75% of all invasive breast cancers.
"Invasive ductal carcinoma is the largest group of malignant mammary tumors, comprising 75% to 80% of mammary carcinomas"
The prognosis of invasive breast cancer is strongly influenced by the stage of the disease, or to what extent the cancer has spread when it is first diagnosed.
Staging takes into consideration size, local involvement, lymph node status and whether metastatic disease is present.
The higher the stage at diagnosis, the poorer the prognosis.
Invasive lobular carcinoma (ILC) is the invasive form of lobular carcinoma some time called infiltrating lobular carcinoma.
About 10% of all invasive breast cancers are invasive lobular carcinomas.
Large studies have shown that approximately 93% of ILC cases are estrogen receptor (ER) positive.
Classic invasive lobular carcinoma (ILC) is characterized histologically by small, discohesive carcinoma cells that infiltrate breast stroma as single cells and in linear files.
"About 10% of all invasive breast cancers are invasive lobular carcinomas"
ILC cells have characteristic plasmacytoid morphology and may also have intracytoplasmic vacuoles and/or signet ring forms.
Morphologic mimics of ILC include both carcinomatous and non-carcinomatous neoplasms, many of which are rarely seen in the breast.

Breast Cancer Hormone Receptor Status
Steroid hormone receptors have important role in the normal development of breast.
Estrogen receptor (ER), progesterone receptor (PR) and human epidermal receptor 2 (Her2/neu) and androgen receptor (AR) are the main steroid receptors which play important role in initiation and progression of the breast cancer.
At puberty 10% mammary epithelial cells expresses ER while 20% cells expresses androgen receptor (AR).
"ER positive breast cancers expresses the ER in their cells sometime these cancers also expresses PR, Her2/neu and AR"
Normally mammary tissue cells expresses the estrogen receptor in less than 1% cells which are essential for the normal development and differentiation of the terminal ductal cells to milk producing cuboidal cells while in breast cancer the level of estrogen receptor (ER-α) is very high.
Depending upon the expression level of ER in breast cancer tissue breast cancer is classified as:
ER positive Breast cancer (ER +Ve): ER positive breast cancers expresses the ER in their cells sometime these cancers also expresses PR, Her2/neu and AR.
ER positive BC is most common type of breast which accounts for 70-80% all types of breast cancer.
ER positive breast cancers are non invasive tumor which is favorable for its management these tumors are associated with low tumor burden and high survival, good prognosis.
Endocrine based therapies are most effective therapies for ER positive BCs.
"ER positive BC is most common type of breast which accounts for 70-80% all types of breast cancer"
Tamoxifen, a ER antagonist specifically target the ER signaling is the most effective drug for the ER positive breast cancers while it not so effective in ER negative breast cancer cases and associated with hair loss, pain in joints and abnormal menstrual cycle and nausea and vomiting.
ER negative Breast cancer (ER -Ve): ER negative breast cells expresses very low level of ER in their cells.
ER-Ve is aggressive type which has highly invasive and has great potential to get metastases.
ER-Ve BCs account for 20-30 % of all types breast cancers. ER –Ve breast cancer associated with poor prognosis, low survivability, high recurrence and high cost of management.
ER negative breast cancer is further sub classified as triple negative breast cancer, basal like breast cancer and apocrine breast cancers.
Triple negative breast cancer (TNBC) is negative for all the three steroid receptors (ER, PR and Her2/neu) which account 10-15 % of all breast cancers among women.
Triple negative breast cancer are most dangerous and highly aggressive type of tumor with greater invasive and metastatic potential.
"ER –Ve breast cancer associated with poor prognosis, low survivability, high recurrence and high cost of management"
Molecular apocrine breast cancer which is negative for ER and PR and Her2/neu and positive for AR had phenotypic characteristics similar to that of ER positive breast cancer.
Molecular apocrine tumor also expresses GHR, EGFR receptors and PRLR.
These tumors account for 4-14% of all types of breast cancers.
Molecular apocrine breast cancer further sub classified as luminal (ER + AR +), apocrine (ER-AR +) and basal (ER- AR-).

Depending upon the expression profile of the steroid hormone receptors and erythroblastosis oncogene B2 (ERBb2 or Her2/neu) breast cancer is classified as Basal like, Luminal A, Luminal B and Her2/neu rich breast cancer.
Basal like tumor are ER, PR and Her2/neu negative and expresses one or more cytokeratin markers 5/6, cytokeratin 17, cytokeratin 14 and EGFR and account 10-15% of all breast cancer.
"Triple negative breast cancer are most dangerous and highly aggressive type of tumor with greater invasive and metastatic potential"
Most of the basal like tumor are IDC-NST type with high tumor grade and triple negative phenotype.
71% of triple-negative cancers were of basal-like subtype and only 77% of molecular basal like tumors were triple-negative.
Luminal A breast cancer is ER, PR positive and Her2/neu negative breast cancer while Luminal B is ER, PR and Her2/neu positive.
Luminal B and Luminal A are hormone receptor positive breast cancer which accounts 70% of all breast cancers.
Inflammatory breast cancer (IBC) is a lethal form of breast cancer associated with inflammation of breast tissue which accounts 1-5% of all breast cancer.
Inflammatory breast cancer is type of invasive ductal carcinoma in which duct is filled with fluid due to blockage of duct result in inflammation.
"Inflammatory breast cancer is a lethal form of breast cancer associated with inflammation of breast tissue which accounts 1-5% of all breast cancer"
IBC characterised by inflammation, redness which covers more than 30% of the total mammary gland and “peau d’orange” appearance and at molecular level by loss of WNT1-inducible-signaling pathway protein 3 (WISP3) signalling and increased expression of E-cadherins and C-guanosine triphosphatase (RhoCGTPase).
Most of the IBC are of grade IV. de Andrés PJ et. al 2013 reported increased level of cytokines IL-6, IL-8 and IL-10 play important role in the inflammatory breast cancers.
With the use of neoadjuvant therapies for the IBC patient survival rate had increased from 30% to 42% and in 70-80% in non IBC patients.
Male breast cancer (mBC) is a rarest type of mammary tumor among male is male breast cancer (mBC) which account ∼1% of all cancer in men.
Hormone receptor positivity (70-90 %) is more in mBC than FBC (60-70%) and lower Her2 over expression.
Biologically mBC differ from FBC and showed lower metastatic tumor profile and chemotherapy, Endocrine therapy and trastuzumab are effective and safe for mBC.
TNM Staging and grading system of Breast cancer
The severnity of the disease breast cancer is grouped into differnt stage diseases in term of its tumor size, number lymph nodes positive for the breast cancer cells, whether breasst cancer cells get metastasses or not metastatic.
"TNM is a standard way of representing the tumor stage and determining tumor grades"
TNM is a standard way of representing the tumor stage and determining tumor grades.
T represent the size of tumor when the size of tumor is less than 1cm the T is represented as T1 which further specified into different category as T1mi, T1a, T1b and T1c as shown in the figure. when tumor size range 2-5 cm it is represented as T2 and when it is more than 5 cm it is represented as T3.
Any size of tumor which as invade to the breast bone is represented as T4 which is further sub categorized as shown in the figure.
The number of nodes positive for the breast cancer cells is represented in terms of N.
When breast cancer cells are get metastases to distant organs from their parental origin it is represented as M1.
Finally the grades of the tumor are determine as according to the TNM status.
Share
-

Origin of Breast Cancer : Breast Cancer...
Continue Reading"Breast Cancer is caused by uncontrolled cell division of the abnormal cells"
What Is Breast Cancer?
Although we have developed lots of understanding about the cancer biology and many therapeutic drugs has been discovered for the treatment of cancers.
We can manage few cancers to some extent such as blood cancer and cervical cancer if detected at early stages.
Still we have big challenge for the development of effective and safe drugs for most of the other cancers. The war against cancer is still challenging.

Cancer is caused by uncontrolled cell division of the abnormal cells. Cancer causing agents are called carcinogens such as chemical carcinogenic (nitrobenzene), DNA damaging radiations (UV raditions) and biological carcinogens (alfa toxin, tumor viruses).
Cancer cells grow and proliferate in defiance of normal controls and able to invade the surrounding tissues and colonise in the distant organs (Metastasis).
Normal cells stop cell division when no more cells are required and undergoes programmed cell death (apoptosis) when it face un-repairable DNA damage while cancer cells do not arrest cell division when there is no more cells are required and becomes resistant to undergo apoptosis.
Most of the cancers arises from a single normal cells by experiencing initial mutation and become abnormal further their progeny cells accumulates additional genetic and epigenetic changes which give them additive advantages to become full blown cancerous cells.
"The war against breast cancer is still challenging"
Development of cancer requires gradual mutational accumulation in various genes- different for different cancers. At each step cancer acquires additional changes to counteracting new challenges during their progression.
Stromal cells evolved along with cancer progression to support the tumour growth. Cancer cells starts secrete some soluble factor which stimulates stromal cells to support the tumor growth.
"Breast cancer is the cancer of mammary gland due to uncontrolled cells division of mammary epithelial cells of the lobules or milk duct of the gland"
Breast is a mammary gland which is mainly meant for the lactation for baby feeding. Breast is glandular fatty connective tissues consist of lobules and ducts.
The basic unit of the mammary gland is alveoli which is lined by milk secreting cuboidal cells and surrounded by myoepithelial cells and these alveoli joined to form lobules which open into the nipple to drain the milk via lactiferous duct.
At puberty when the level of steroid hormones gets increased induced the division of epithelial cells of lobules and ducts.
Fatty connective tissues surround lobules and ducts, protect and give shape to breast.
Oestrogens are among most important hormone to which breast tissues respond under the influence of estrogens the epithelial cells divide in a controlled manner when the level of this hormones abnormal epithelial cells divide in uncontrolled manner and further undergoes some additional changes which leads to development of breast cancer.
Breast cancer is the cancer of mammary gland due to uncontrolled cells division of mammary epithelial cells of the lobules or milk duct of the gland.
Uncontrolled cell division in the fibroblast of the breast stromal tissue is called sarcoma.
"Accumulation of genetic abnormalities, over time, leads to development of a colony of cells which acquired the proliferation potential and outpace the surrounding cells"
Origin Of Breast Cancer
Like most of the cancers breast cancer is also arises from the single abnormal cell which experience initial mutations and start proliferating abnormally and exponentially result in ductal hyper-proliferation which lead to in situ and invasive carcinoma and subsequently get metastases.
As a result of accumulation of large number of individual genetic mutations complex internal signalling system of a cell get altered.
When assembled in a single breast cell, disrupt the control system to the extent that the cell functions autonomously in an erratic and irregular manner.
Continual replication of a corrupted cell results in the formation of a colony of abnormal cells that may accumulate other additional aberrant mutations which gives an additive advantage for the proliferation of abnormal cells.

Both genetic and epigenetic changes are responsible for the cell to become cancerous.
Genetic changes are the resultant of sequence of random, accidental, spontaneous mutations during normal stem cell replication.
Genetic changes are get repaired by DNA damages repair system of the cells.
When the defence system is corrupted, cells carrying the abnormal genes are able to avoid elimination, eventually passing abnormal mutations from one cell generation to the next.
Accumulation of genetic abnormalities, over time, leads to development of a colony of cells which acquired the proliferation potential and outpace the surrounding cells.
Previous studies suggested role of steroid hormones in the induction of the genetic mutation in vitro cultured stem cells these steroid hormones also increases the rate occurrence of genetic mutation in vitro conditions.
"Cytokines secreted by resident or infiltrating cells of the microenvironment either support or inhibits the tumor growth"
Tumor micro-environment also play important role in the progression of the breast cancer.
Breast cancer tumor microenvironment consist of fibroblast, adipocytes epithelial cells, stromal cells and infiltrating cells.
Cytokines secreted by resident or infiltrating cells of the microenvironment either support or inhibits the tumor growth.
Cytokines are small molecular proteins which required in picomolar concentration for their biological activity.
Cytokines are important component of the host defence system mainly responsible for the intercellular communication.

Some cytokines have immune suppressive effect to suppress host immune response after clearance of foreign pathogen.
Cancer cells start secreting these tumor suppressive cytokines to suppress the host immune response against cancer cells and thus these cytokines support the tumor growth alternatively.
"Genetic changes are get repaired by DNA damages repair system of the cells"
Steroid receptor are important determinant for the deciding the treatment for the breast cancer.
Estrogen receptor(ER), progesterone receptor (PR), herceptin receptor (Her2/neu).
Estrogen receptor is most frequently expressed in 70-80% breast cancer while progesterone receptor expressed in 50-70% of the breast cancer.
Androgen receptor, a steroid receptor also play important role in progression of breast cancer.
Several studies reported androgen receptor expressed in 70-90% breast cancer cases however, in some sub types of breast cancer AR receptor positivity associated with poor prognosis while in other sub types it is associated with good prognosis and increased cell overall survival suggest that complex role of androgen in breast cancer.
However, there are debatable study out there about the role of different cytokines in breast cancer and also response of breast cancer cells with different cytokines and androgen receptor and their target genes. In addition, effect of androgen (DHT) and SARM on cytokines profile and on cytokine signalling in breast cancer cells in not well characterized.
Share
-

Mammalian Cell Transfection: Overview of Transfection Methods
Continue ReadingWhat is Transfection?
Transfection is the process by which foreign nucleic acids (DNA or RNA) introduced into eukaryotic cells by non-viral methods.
Using various physical or chemical techniques, DNA or RNA transfer technology enables the study of gene function and protein expression in a cellular environment.
Principle of Transfection
TurboFect in vitro Transfection Reagent is a sterile solution of a proprietary cationic polymer in water.
The cationic polymer forms stable, positively charged complexes with negatively charged DNA.
These complexes protect DNA from degradation and facilitate gene delivery into eukaryotic cells.
TurboFect is ideal for transfection of a variety of cells, such as adhernt cells, suspension cells, including primary and difficult-to-transfect cells.
It can be performed in the presence or absence of serum.
TurboFect demonstrates superior transfection efficiency and minimal toxicity when compared to lipid-based or other polymer-based transfection reagents.

Created with BioRender
Transfection Requirements
Sterile:
1. TurboFect in vitro Transfection Reagent
2. 6 well plates
3. Microtips
4. Growth medium
5. Cells (A549 Cell line)
6. Trypsin EDTA
7. DNA
Non sterile:
8. 70% IPA
9. Cotton balls
Transfection Procedure
1. Prior to the day of transfection, 5 x 104 adherent cells were plated in each well in a 12 well plate containing 1 ml of growth medium.
2. 1μg of DNA was diluted in 100μl of serum free DMEM or RPMI media
3. 2μl of turbofect reagent was added to the diluted DNA (2μl/1μg of DNA) and mixed immediately by pipetting or vortexing and it was kept for incubation for 15-20 minutes at room temperature
4. 100μl of this turbofect/DNA mixture was added dropwise to each well without removing growth medium from the cells
5. Plate was gently rocked to achieve even distribution of the complexes.
6. It was further incubated at 37 ̊C in CO2 incubator
7. After 24 hours, treatment with selection antibiotic (Geneticin) was done at concentration of 400μg/ml.
Citations:
Share
Similar Post:
-
Cellular Drug Uptake with Fluorescent Sensor
Continue ReadingWhat is Cellular Drug Uptake?
A key function of macrophages is to engage and clear invading microorganisms.
A variety of receptors present on the plasma membrane of phagocytes mediate interactions with pathogens.
A novel formulation of nanoparticles/ drugs needs to be checked for the uptake by macrophages.
Macrophages are involved in evading the foreign particles and eliciting the immune response.
Thus, in vitro drug uptake is necessary before the formulation can move ahead and be used for in vivo studies.
Depending on the type of molecule and the mode of its uptake, endocytic process has been broadly classified into pinocytic, phagocytic and receptor mediated pathways.
However, further categorization of the processes has been done depending on whether or not the process utilizes clathrin, a molecule known to be very important for transport of transmembrane receptors involving coated pits.
Phagocytosis is engulfment of large particulate matter specially shown by macrophages and neutrophils.
The engulfment of particles in this process forms phagosomes. The vesicles in this case are usually larger than 250nm.
Similarly, pinocytosis is a process of “cell drinking”, here the particles taken up by the cell are usually very small in size.
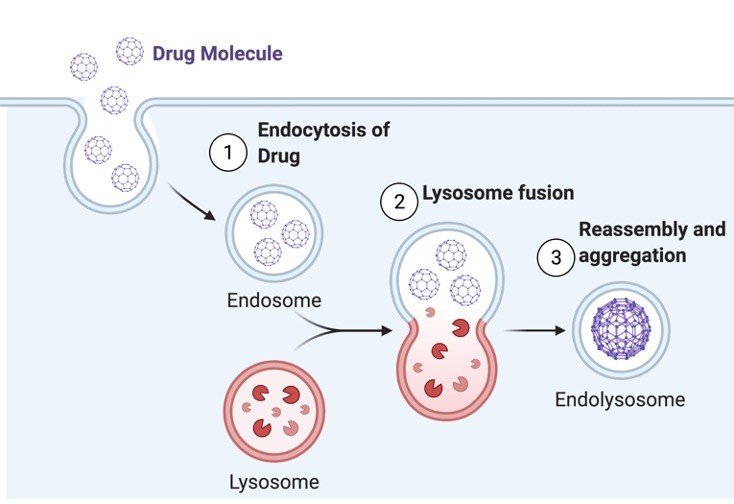
Created with BioRender
About Current Cellular Drug Uptake Protocol
In the present experiment, we would study about uptake of fluorescent beads which are opsonized with IgG.
These beads are ~2μm in diameter; hence they would be taken up by phagocytosis route.
Opsonization with IgG enhances their phagocytic uptake.
Along with this formulation of iron particles coated with RBITC-BSA (Rhodamine B Isothiocyanate-Bovine Serum Albumin) would also be studied.
Since iron particles are very small is size ~25nm, they are usually taken up by pinocytosis/receptor mediated endocytosis.
To understand the mechanism of uptaking particles it needs to be checked at 37°C versus 4°C, competitive inhibition and also a saturation curve of receptors, which can give us the idea whether the uptake is receptor mediated or not.
Principle of Cellular Drug Uptake
Isolated rat peritoneal macrophages are used for in vitro drug uptake of particles which are in the range of nanometer to micrometers.
The particles which are in the range of nanometers (~25nm) are expected to be internalized by the process of pinocytosis/receptor mediated endocytosis, whereas those in micrometer (2μm) range are internalized by the process of phagocytosis.
Cellular Drug Uptake Requirement
1. Rhodamine B Isothiocyanate-Bovine Serum Albumin coated iron particles.
2. Opsonized fluorescent beads
3. Phosphate buffer saline
Procedure of Cellular Drug Uptake
1. Visualize rat peritoneal macrophages isolated from earlier experiment in inverted microscope. Assess for their morphology (oval, translucent) and size.
2. For opsonization of fluorescent beads, add 50 μl of beads suspension in 100 μl of rabbit serum. Put the mixture at 37 C shaker for two hours.
3. Treat the macrophages with RBITC-BSA-iron and opsonized Fluorescent beads.
4. After the treatment for respective duration, wash with Phosphate buffer saline for 3 times.
5. Add 500μl of PBS in each well/petridish and visualize in fluorescent microscopy.
Cellular Drug Uptake Analysis
Drug uptake could be studied quantitatively as well as semi-quantitatively.
To study the uptake quantitatively, after thorough washing, we need to scrap out all the cells, lyse them using hypotonic lysis buffer and then measure the fluorescence using fluorimeter.
More percent of drug uptaken by macrophages correlate to more fluorescence in lysed macrophages.
Evaluate results of the percentage of drug uptaken by comparing the fluorescent drug inside the cell per milligram of protein versus drug suspension added.
Semiquantitative estimation can be done using fluorescent microscope.
Checking out the fluorescent images as well as phase contrast gives us understanding of drug uptake inside the cell.
Internalization of drug inside the macrophages will form vesicles (endosomes).
Thus, more is the uptake of the drug per cell; we will visualize more endosomes per cell.
We can calculate the percentage of phagocytes out of the total number of cells with fluorescent phagocytic vesicles to assess the phagocytic uptake.
Assessment of florescent beads is to be done in blue filter and for nanoparticles in green filter using florescent microscope.
Cellular Drug Uptake Citations:
Share
Similar Post:
-

Primary Cell Culture: Isolation and Culture of...
Continue ReadingWhat is Primary Cell Culture
Primary cultures denote those cultures which consist of cells before subculturing.
It represents the mixture of cell types present in a given tissue and hence they are closer to the tissue architecture in vivo.
In primary cultures the tissue pieces can be grown as explants cultures or tissues can be subjected to enzyme treatment and dissociated cells are grown as monolayers.
Distinguishing Features of Organ Cell Culture
Architecture of tissues in vivo is retained.
Their functionality is also retained, since they maintain their differentiated character.
The tissue is cultured at the liquid-gas interface.
Grids, gels and sponge matrix which maintain 3D structure of organs are used as substrates.
Attempts are made to deliberately minimize proliferation and migration of cells.
Advantages of Organ Cell Culture
Organ cultures are used for studies involving cell interactions, functional properties of cells, effects of external agents or effects of products of one organ on another.
Characterization of these cultures can be easily carried out from histological studies.
Biochemical differentiation is also possible.
Disadvantages of Organ Cell Culture
1. Organ cultures cannot be propagated for long periods.
2. Each experiment requires a new culture.
3. Quantitation of results is very difficult.
4. High inter-sample variation is required for reproducibility and homogeneity of results.
Requirements of Organ Cell Culture
Unsterile-fertilized chick eggs incubated for 10 days.
Sterile-surgical instruments, 65 mm pertidishes, PBS-A with antibiotics, 35 mm petridishes, RPMI 1640 with 20% FCS, trypsin 50 ml conical flasks, magnetic bars.
Organ Cell Culture Procedure
1. Wipe precleaned eggs with savlon and iodine.
2. Hold the egg with broad side up and tap the shell gently with scalpel to crack it uniformly all around.
3. With a sterile forceps gently remove the shell pieces and displace the CAM to expose the embryo.
4. Lift up the embryo by inserting one arm of a blunt forceps below the neck of the embryo and transfer it to a petridish with PBS-A.
5. Wash the embryo twice with PBS-A, decapitate and wash again twice.
6. Dissect out all the visceral organs wash the remaining parts of the embryo with PBS-A, mince it with sharp scissors and transfer to a 50 ml conical flask.
7. Add 10-15 ml trypsin, put a magnetic bar in the flask and keep the flask on the magnetic stirrer with a gentle speed to avoid frothing for 30 mins.
8. Remove the flask from the stirrer, allow the pieces to settle, transfer the supernantant to a centrifuge tube and centrifuge for 10 mins at 1500 rpm.
9. If necessary, repeat the step of trypsinization once or twice more.
10. Pool all the cell suspensions, make a cell count and add about 1 X 10^5 cells/ml in 35 mm petri dishes and incubate in the CO2 incubator.
Observation: Within 24 hrs cells form monolayer and show semi confluency in 4-5 days.
Organ Cell Culture Citations
Share
Similar Post:
-
Primary Cell Culture: Isolation of Rat Peritoneal...
Continue ReadingAdvantages of Primary Cell Culture
Primary culture is the culture of cells directly obtained from animal tissues.
It has the following advantages:
More in vivo like characteristics
Cells can be obtained from rare or unusual species
Patient derived tissues can be utilized for direct experimentation
Cells that are not available as cell lines
About Rat Peritoneal Macrophages Cell Culture
The two most convenient sources of primary murine macrophages are the bone marrow and the peritoneal cavity.
Resident peritoneal macrophages can readily be harvested from mice and purified by adherence to tissue culture plastic.
The injection of thioglycollate broth into the peritoneal cavity produces an inflammatory response allowing the purification of large numbers of elicited macrophages.
The macrophages obtained by the following protocol are normally 90% pure and can be used further for other assays like drug uptake, phagocytosis etc.
Cell Culture Requirements
Sterile:
1. Phosphate Buffer Saline (PBS) (i.e. 137mM NaCl, 2.7mM KCl, 10mM Na2HPO4.2H2O and 2 mM KH2PO4, pH 7.4).
2. Geys solution: Stock A (0.65M NH4Cl, 0.024 M KCl, 10.56 mM Na2HPO4, 0.89 KH2PO4, 0.02 M Glucose, 0.141 mM Phenol Red); Stock B (0.207 mM MgCl2.6H2O, 0.057 mM MgSO4.7H2O, 0.306 mM CaCl2); Stock C (2.68 mM NaHCO3); Geys Solution (Make Fresh): 20 Parts of Stock A 5 Parts of Stock B 5 Parts of Stock C 70 Parts of autoclaved deionized water.
3. RPMI medium supplemented with 10% FBS and antibiotics
4. FBS
5. 23G and 18G needles
6. 10ml syringe
7. 3% w/v Brewer’s complete thioglycollate broth: 3g dissolved in water, Autoclave and store in dark and should be allowed to age for atleast 15 days before using for the experiment
8. Scissors and forceps
Non-sterile:
9. 70% v/v isopropyl alcohol or ethanol
10. Cotton
11. Ice
Cell Culture Procedure
1. Inject the rat with 5 ml aged thioglycolate medium in the peritoneal cavity using a syringe with 27 G needle.
2. After 72 hr and immediately before harvesting the macrophages, sacrifice the rat by CO2 asphyxiation.
3. Disinfect all the external areas of the rat with 70% IPA.
4. Make a small incision in the abdominal region and gently rip the skin downward to expose the intra-peritoneal cavity.
5. Inject 10 ml of sterile PBS into the intra-peritoneal cavity using a syringe with a 23 G needle and massage the abdomen gently for 2 min.
6. Retract the injected PBS using a syringe with 18 G needle. Care should be taken not to injure the internal organs during this process.
7. Centrifuge the Macrophage suspension at 1300 rpm, 7 min and 40C and observe the pellet.
Note: If RBCs are visualized, then add 1ml of complete Gey’s solution and incubate on ice for 2-5 minutes. Gently overlay 1ml of FBS to this suspension and spin at 300g for 10 minutes at 4oC. Wash once with PBS containing 5% FBS and then resuspend in 1 ml of RPMI supplemented with FCS and antibiotics.
8. Count the cells using hemocytometer
9. Plate the resident peritoneal cells RPMI culture medium at 3 x 10^5 cells per well in a 24-well tissue culture plate or 106 cells in 30mm sterile petridish and incubate for 60 mins at 37°C.
10. Remove the nonadherent cells by washing 3 times in 500ml warm PBS, using a gentle swirling action.
11. The adherent cells normally contain >90% macrophages
12. These macrophages will be used for drug uptake assay
Primary Cell Culture Citations:
Share
Similar Post: